Background:
Mastocytosis is a heterogenous disease of mast cell (MC) clonal proliferation with pathologic accumulation in organ systems such as but not limited to skin, bone marrow, gastrointestinal tract, liver and spleen. Mast cells are CD34+, CD117+ (stem cell factor, KIT) with greater than 90% of people with mastocytosis harboring KIT D816V activating mutations which allows for increased proliferation and accumulation of neoplastic mast cells leading to downstream mast cell activation and intolerable symptoms.
Advanced systemic mastocytosis (advSM) is categorized into three major subtypes, aggressive SM, MC leukemia and SM with an associated hematologic neoplasm (SM-AHN). SM-AHN is the most common advSM subtype accounting for about 75% of advSM cases and typically includes myeloid neoplasms such as MDS, CMML, or MPN. In most pts with SM-AHN, clinical symptoms is driven by the MC component and the goal of treatment with KIT inhibitors is to decrease the MC burden, thus improving quality of life and overall survival (Nat Med refs), However, the median survival of SM-AHN remains poor at 2-3.5 years with many pts transforming to advanced myeloid neoplasms especially an acute myeloid leukemia (AML).
The use of next generation sequencing has helped to prognosticate patients with AdvSM. Patients with advSM typically exhibit multiple somatic mutations other than KIT D816V We describe the molecular signature of patients with SM-AHN who transformed to AML. Commonly involved genes are TET2, SRSF2, ASXL1, RUNX1, and CBL with inferior OS and adverse clinical characteristic associated with SRSF2/ ASXL1/ RUNX1 (S/A/R). To better prognosticate, Jawhar and colleagues introduced the MARS score. This incorporated molecular findings in AdvSM to help estimate overall survival which identified 3 risk categories (low, intermediate, and high). The overall survival in low, intermediate and high were not reached, 4.3 years, and 1.9 respectively for patients with AdvSM. While mutational findings in the MARS score can help prognosticate overall survival in these patients, the molecular footprint of progression or transformation to acute myeloid leukemia has not been established. We describe the molecular signature of patients with SM-AHN who transformed to AML.
Methods: We analyzed 10 pts with SM-AHN who transformed to AML from 2000-2022. Of those patients, 7 were found to have full next generation sequencing at the time of initial consultation and transformation. These data were collected and descriptive analysis was performed.
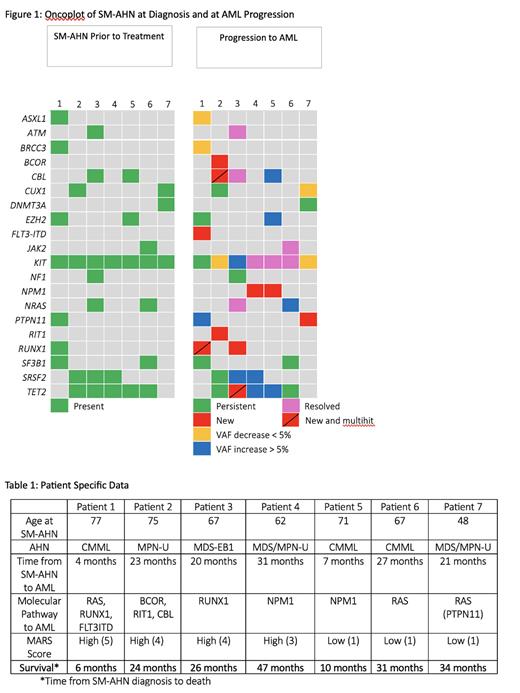
Results: We identified 7 pts with SM-AHN who progressed to AML. At SM-AHN diagnosis all pts had at least 2 additional co-mutations with a maximum of 7 co-mutations along with KIT D817V. The most common co-mutations were splicing factor mutations ( SRSF2 and SF3B1), TET2, RAS EZH2, CUX1 and CBL. Six of the seven received treatment for SM-AHN. These patients received treatment with multikinase inhibitors or hypomethylating agents.
The acquired driver mutations were NPM1, FLT3-ITD, RUNX1, and RAS pathway. In patient 1, a new FLT3-ITD and RUNX1 was acquired with an increased VAF of a RAS pathway mutation ( PTPN1) which was originally present in the SM-AHN diagnosis. Patient 3 also developed a new RUNX1 mutation at AML transformation. Patients 4 and 5 had new NPM1 mutations and patients 6 and 7 had a new RAS pathway mutations . Patient 2 acquired new BCOR, RIT1 and CBL mutations. Upon transformation to AML, 5/7 patients had resolution or a decrease of greater than 10% of KIT D816V VAF. One of the seven had no change in KIT D816V VAF at the time of transformation, this was also the only patient to receive no prior therapy. Only 1/7 had a VAF increase of greater than 20% in KIT D817V at transformation.
The MARS score was calculated at the time of diagnosis of SM-AHN which showed that only 4 of 7 had a high MARS score. Time from SM-AHN diagnosis to AML progression ranged from 4-31 months. Survival was calculated from time of SM-AHN diagnosis to death which ranged from 6-47 months.
Conclusion: Acquisition of known AML driver mutations (RAS, RUNX1, NPM1, FLT3-ITD) are commonly found in SM-AHN patients at time of progression to AML. The MARS score calculated in our patients did not correlate with time to transition to AML.
Disclosures
Luskin:Novartis: Research Funding; Pfizer: Honoraria; Novartis: Honoraria; Jazz: Honoraria; AbbVie: Research Funding. Stahl:Novartis: Membership on an entity's Board of Directors or advisory committees, Other: GME activity ; Kymera: Membership on an entity's Board of Directors or advisory committees; Boston Consulting: Consultancy; Sierra Oncology: Membership on an entity's Board of Directors or advisory committees; Dedham group: Consultancy; GSK: Membership on an entity's Board of Directors or advisory committees; Clinical care options: Other: GME activity ; Rigel: Membership on an entity's Board of Directors or advisory committees; Haymarket Media: Other: GME activity ; Curis Oncology: Other: GME activity . Lindsley:Sarepta Therapuetics: Consultancy; Verve Therapuetics: Consultancy; Jazz Pharmaceuticals: Consultancy; Vertex Pharmaceuticals: Consultancy; Qiagen: Consultancy; Bluebird bio: Consultancy, Membership on an entity's Board of Directors or advisory committees; Takeda Pharmaceuticals: Consultancy. DeAngelo:GlycoMimetics: Research Funding; Autolus: Honoraria; Takeda: Honoraria; Pfizer: Honoraria; Novartis: Research Funding; AbbVie: Research Funding; Blueprint: Research Funding; Jazz: Honoraria; Novartis: Honoraria; Kite: Honoraria; Servier: Honoraria; Amgen: Honoraria; Incyte: Honoraria; Blueprint: Honoraria; Gilead: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal